UV-C Light Technology Explained: How It Kills Bacteria and Viruses

If you've recently looked into water purifiers, air sanitizers, or hospital-grade cleaning equipment, you've probably encountered the term UV-C light. Manufacturers claim it can destroy bacteria, viruses, and other microorganisms without chemicals — and for once, the science actually backs them up. But understanding how UV-C light kills bacteria requires a brief detour into photochemistry, because the mechanism is more precise — and more fascinating — than most people realize.
This guide breaks down the ultraviolet spectrum, explains the exact molecular process by which UV-C radiation disables pathogens, reviews the research on its effectiveness, and discusses the real-world limitations you need to know before trusting any device that uses it.
The Ultraviolet Spectrum: UV-A, UV-B, and UV-C
Ultraviolet light sits just beyond the visible spectrum, at wavelengths shorter than violet light (roughly 10–400 nanometers). Most people know that UV radiation from the sun can tan or burn skin, but the full UV spectrum is divided into three distinct bands — and they behave very differently from one another.
UV-A (315–400 nm) is the longest-wavelength, lowest-energy band. It makes up about 95% of the UV radiation that reaches Earth's surface. UV-A penetrates deep into skin tissue and is responsible for tanning, photoaging, and contributing to certain skin cancers. It has very little germicidal power because its photons don't carry enough energy to efficiently break chemical bonds in DNA.
UV-B (280–315 nm) is partially filtered by the ozone layer. It carries more energy than UV-A and is the primary cause of sunburn. UV-B has some germicidal effect, but its effectiveness against pathogens is inconsistent compared to UV-C.
UV-C (100–280 nm) is the shortest-wavelength, highest-energy band — and it is almost entirely absorbed by Earth's atmosphere before reaching the surface. Natural sunlight at ground level contains essentially no UV-C. This is precisely why UV-C light disinfection science is so valuable: microorganisms on Earth have had almost no evolutionary pressure to develop defenses against it.
The Science of UV-C Germicidal Irradiation
The mechanism behind how UV-C light kills bacteria operates at the molecular level, targeting the genetic material that every living cell — and every virus — depends on to function and reproduce.
Why 253.7 nm Is the Critical Wavelength
Germicidal UV lamps are typically engineered to emit light at or near 253.7 nanometers. This isn't an arbitrary choice. DNA and RNA are composed of nucleotide bases — adenine, thymine, cytosine, and guanine (with uracil replacing thymine in RNA). These bases have a strong absorption peak at approximately 254 nm. In other words, photons at this wavelength are absorbed by the nucleobases with exceptional efficiency, maximizing the energy transfer that triggers damage.
This peak absorption is what makes ultraviolet germicidal irradiation (UVGI) so effective: the light essentially targets the exact molecular structures that make a microorganism dangerous.
Thymine Dimer Formation
When a UV-C photon is absorbed by two adjacent thymine bases on the same DNA strand, a covalent bond forms between them, creating what is called a cyclobutane pyrimidine dimer — commonly referred to as a thymine dimer. This is the primary form of damage UV-C induces.
A thymine dimer is essentially a structural kink or "knot" in the DNA helix. When the cell attempts to replicate its DNA before dividing, the replication machinery (DNA polymerase) stalls or misreads the damaged sequence. The result is one of three outcomes: the cell cannot divide at all, it produces a defective copy that cannot function, or it triggers its own programmed death (apoptosis in eukaryotic cells). In viruses, the damage to RNA or DNA prevents the virus from hijacking host cell machinery — it simply cannot replicate.
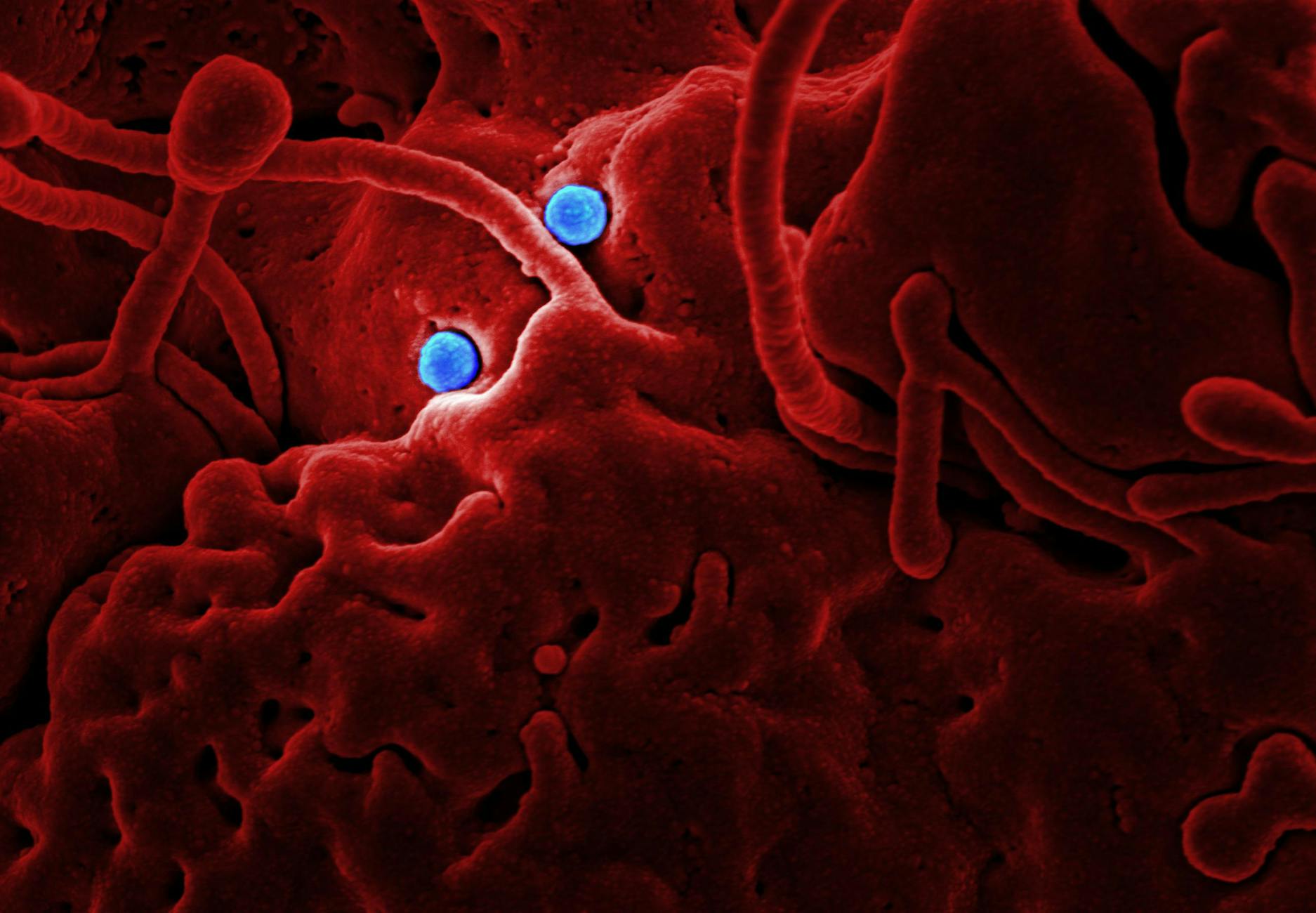
Why Microorganisms Cannot Simply Repair the Damage
A fair question: don't cells have DNA repair mechanisms? Yes — and UV-C exploits an important limitation of those mechanisms. While many organisms possess a repair pathway called nucleotide excision repair (NER), which can cut out and replace damaged thymine dimers, this process has a capacity limit. A sufficiently high UV-C dose creates so many simultaneous lesions throughout the genome that the repair machinery is overwhelmed. The organism accumulates damage faster than it can fix it.
Furthermore, viruses generally lack their own DNA repair systems entirely — they depend on host cell machinery, which is not optimized for this purpose. Single-celled organisms like bacteria also tend to have less robust repair capacity than multicellular organisms, making them particularly vulnerable.
The key metric here is UV dose, measured in millijoules per square centimeter (mJ/cm²). Different pathogens require different doses to achieve a 90% reduction (1-log kill) or a 99.99% reduction (4-log kill). For example, research published in the Journal of Applied Microbiology has shown that Escherichia coli requires only about 3–6 mJ/cm² for a 4-log reduction, while more resistant organisms like Bacillus subtilis spores may require 80–100 mJ/cm² for the same result.
UV-C Light Disinfection Science: Effectiveness Against Pathogens
The body of peer-reviewed research on UV-C light disinfection science is extensive, spanning several decades. Here is what the evidence shows across major pathogen categories.
Bacteria: UV-C is highly effective against a broad spectrum of bacteria, including antibiotic-resistant strains. A landmark study by Böhm et al. (2016) in PLOS ONE demonstrated effective inactivation of methicillin-resistant Staphylococcus aureus (MRSA) at doses achievable by commercial UV-C systems. Salmonella, E. coli, and Listeria are all similarly susceptible.
Viruses: Both enveloped viruses (like influenza and coronaviruses) and non-enveloped viruses respond to UV-C irradiation. Research published in the American Journal of Infection Control found that far-UVC light (a specific subset near 222 nm) effectively inactivated aerosolized influenza viruses. Studies following the emergence of SARS-CoV-2 confirmed that the novel coronavirus was inactivated by UV-C doses consistent with prior coronavirus research.
Fungi and Mold: Fungal spores are generally more resistant than bacteria or viruses due to their protective outer layers. Higher UV doses (often 100–200 mJ/cm²) are required for effective inactivation. UV-C is still used in HVAC systems to prevent mold colonization, particularly in humid environments, though it performs best as a preventative measure rather than a remediation tool once heavy mold growth is established.
Real-World Applications of Ultraviolet Germicidal Irradiation
Hospital and Healthcare Settings
Hospitals have used ultraviolet germicidal irradiation since the 1930s to reduce airborne transmission of tuberculosis and other pathogens in surgical suites and patient wards. Modern systems include pulsed xenon UV robots and ceiling-mounted UV-C fixtures that disinfect patient rooms between occupancies. A 2017 study in Infection Control & Hospital Epidemiology found that adding UV-C disinfection to standard terminal cleaning protocols reduced the rate of healthcare-associated infections caused by target organisms by approximately 30%.
Water Treatment
Municipal water treatment facilities have adopted UV-C disinfection as a final treatment stage, particularly to address pathogens like Cryptosporidium and Giardia that are resistant to chlorination. Because water is transparent to UV-C at low turbidity, light can penetrate the flow and inactivate pathogens efficiently without introducing any chemical byproducts. The U.S. Environmental Protection Agency (EPA) recognizes UV-C as an effective treatment technology for drinking water disinfection.
Air Purification and HVAC Systems
UV-C lamps installed in HVAC ductwork or standalone air-handling units irradiate air as it circulates, reducing the concentration of viable airborne pathogens. This application is particularly relevant in enclosed, poorly ventilated spaces where respiratory pathogens can accumulate. Studies on upper-room germicidal UV systems — fixtures mounted near the ceiling that irradiate the upper portion of a room — have demonstrated reductions in airborne Mycobacterium tuberculosis and other infectious aerosols.
Consumer Devices
The consumer market for UV-C devices has expanded significantly, with products ranging from wand-style sanitizers and smartphone sterilization boxes to water bottle purifiers and countertop air purifiers. Devices like these use UV-C light technology in principle, though their real-world effectiveness varies considerably depending on lamp intensity, exposure time, and whether the geometry of the device actually ensures adequate irradiation of all surfaces. A consumer device with a weak lamp or inadequate exposure time may deliver far less UV dose than laboratory-validated professional equipment.
Safety Considerations: Why UV-C Requires Proper Shielding
The same properties that make UV-C lethal to microorganisms also make direct exposure harmful to humans. UV-C radiation is a known human carcinogen (Group 1, International Agency for Research on Cancer) when exposure is chronic or at high intensity. Acute effects include:
- Photokeratitis — essentially a sunburn of the cornea, causing temporary but extremely painful eye inflammation. Even brief exposure to a UV-C source can cause this.
- Erythema — reddening and burning of exposed skin, similar to severe sunburn.
- Potential long-term carcinogenic risk with repeated unprotected exposure.
Professional UV-C systems are engineered with automatic shutoffs, motion sensors, and interlocks that prevent operation when people are present. Consumer devices should be used strictly according to manufacturer instructions, with no direct skin or eye exposure during operation. The conventional germicidal wavelengths around 254 nm are particularly dangerous to the eyes — appropriate UV-protective eyewear is essential for anyone working with UV-C equipment professionally.
One area of active research involves far-UVC light (207–222 nm), which appears to be effectively blocked by the outer, non-living layers of human skin and the tear film of the eye, potentially making it safer for occupied-room use. Studies by David Brenner at Columbia University's Center for Radiological Research have shown promising results for far-UVC as a safer alternative, though regulatory frameworks around its use are still being established.
Limitations of UV-C Disinfection
UV-C is a powerful tool, but it is not a universal solution. Understanding the limitations is essential to applying it correctly.
The shadow problem. UV-C light travels in straight lines. Any surface that is not directly in the line of sight of the lamp is in a "shadow" and receives little to no irradiation. This is critical for surface disinfection — the underside of a phone, the inside of a complex medical device, or anything tucked behind another object will not be adequately disinfected. Professional systems address this with multiple lamp positions, reflective surfaces, or robotic movement.
Organic matter interference. Blood, mucus, soil, and other organic matter on a surface significantly reduce UV-C penetration. In healthcare settings, UV-C is used as a supplement to chemical cleaning, not a replacement for it. Surfaces must first be physically cleaned before UV-C can reliably inactivate residual pathogens.
Distance and the inverse-square law. UV-C intensity drops off rapidly with distance, following the inverse-square law: doubling the distance from the source reduces intensity to one-quarter. A device that delivers an adequate dose at 10 cm may provide insufficient dose at 30 cm.
Exposure time. Achieving a meaningful log reduction requires a specific UV dose, which is a product of intensity and time. Brief passes with a wand sanitizer, for example, may not deliver adequate dose on all areas of a surface, even if the wand emits genuine UV-C light.
Lamp degradation. UV-C lamps lose output over time. A lamp that was effective when new may deliver substantially less irradiance after several thousand hours of use, yet still appear to emit light visibly. Regular lamp replacement or dose verification is important in professional settings.
What Research Shows About UV-C Effectiveness
The scientific consensus on UV light sanitizer effectiveness is clear when conditions are controlled: UV-C at appropriate doses reliably inactivates a wide range of pathogens. The evidence base includes:
- A comprehensive review in Photochemistry and Photobiology (2020) confirmed UVGI efficacy against SARS-CoV-2, noting dose requirements consistent with other betacoronaviruses.
- A meta-analysis in BMC Infectious Diseases found statistically significant reductions in healthcare-associated infections when UV-C terminal room disinfection was added to standard protocols.
- EPA-reviewed data supports UV-C as a validated treatment for drinking water disinfection, with performance standards codified in the EPA's Ultraviolet Disinfection Guidance Manual.
- Research from the National Academies of Sciences has noted that upper-room UVGI can significantly reduce the airborne transmission of respiratory infections in congregate settings.
The critical caveat in all of this research is dose. Studies that show high efficacy are conducted under controlled conditions with verified, calibrated UV-C output. Consumer products claiming germicidal UV-C performance should ideally provide validated dose data from independent testing labs — not just marketing claims.
The Difference Between UV-C vs UV-A and UV-B in Practical Terms
When evaluating any UV device, understanding UV-C vs UV-A UV-B distinctions matters enormously. Many inexpensive devices sold as "UV sanitizers" emit UV-A or UV-B rather than UV-C. These products may produce a characteristic purple-blue glow that looks impressive but provides negligible germicidal action at the wavelengths and doses involved. Genuine UV-C sources should specify a peak emission near 254 nm (for mercury-vapor lamps) or 275 nm (for UV-C LEDs), and ideally include third-party irradiance data.
UV-C LEDs are a newer technology that offers advantages in form factor, lamp life, and the ability to tune wavelength — but currently available UV-C LEDs are significantly less efficient than mercury-vapor lamps in terms of optical power output, which affects their germicidal performance at a given distance and exposure time.
Key Takeaways
UV-C light is one of the most well-validated non-chemical disinfection technologies available. The science is not marketing hype — how UV-C light kills bacteria is understood at the molecular level, replicated in peer-reviewed research across decades, and applied in some of the most rigorous sanitation contexts on Earth, from municipal water supplies to operating rooms.
What matters is dose. A device that emits genuine UV-C at 253.7 nm and delivers the required millijoules per square centimeter to an unobstructed surface will reliably inactivate the pathogens the research says it will. A device that cuts corners on lamp power, exposure time, or coverage geometry will not — regardless of what the packaging claims.
For consumers evaluating UV-C products, the right questions to ask are: Does this emit genuine UV-C (around 254 nm or 275 nm), not UV-A? What is the irradiance at the relevant distance? Is there independent testing data? Does the device geometry actually expose the target surface without shadows? Answering these questions turns "does UV-C work?" from a marketing debate into an engineering problem — one with clear, evidence-based answers.